A. S. Chamove, BA, MA, MiBiol. and J. R. Anderson, BSc.
Stirling University, Psychological Department,
Stirling FK9 4LA, U.K.
SUMMARY
The effects of the presence of woodchips as a floor covering were assessed in two pens housing 28 monkeys. No negative effects were found, while aggression decreased and time foraging on the floor increased. The chips were less expensive, they reduced odour, and the animals appeared more well-adjusted than with no floor covering.
INTRODUCTION
In the wild, non-human primates spend a high proportion of time searching for small food items. When captive, however, our concern with health and hygiene obviates these behaviours. Most authorities recommend that excreta be removed from cages at least once daily whether or not the animals can contact the excreta.1 2 3 This usually results in the recommendation that enclosures or cages be designed so as to be maximally cleanable, and if litter is used, it be restricted to the cleaning tray below mesh floors.4 The rare advocacy of litter with primates is most sensibly illustrated as follows:
"If litter or bedding is used in animal cages or pens, it should be changed as often as necessary to keep the animals dry and clean, and to minimise offensive odours" (p. 6).2
With primates, they continue, daily cleaning may be necessary .
Nevertheless, and against all of this advice, we chose to evaluate woodchips as a long-term deep litter for macaques. We felt one should evaluate the effects on psychological well-being: giving the animals something to occupy their time, giving them something they do naturally such as searching through and manipulating the litter; physical well-being: fighting, food distribution, cleanliness, dryness, disease transmission, noise reduction, and cold surfaces; economy; and aesthetics: odour, visibility of animals, social behaviour and appearance of the animals. It was felt that deep litter might affect all of these aspects of laboratory primate maintenance.
METHODS
Two groups of stumptailed macaques (Macaca arctoides) were used. The Juvenile group consisted of 15 laboratory-born monkeys, five of which were males, and averaging 4-6 years of age. The three youngest members had been living in this pen group for 5 months, all others for at least 15 months. All of these subjects had been separated from their mothers in the first week of life, reared in individual cages (as described by Chamove5,6), but (all except three) had experienced daily social interaction with a minimum of three other monkeys. From this we can expect social behaviour approximating that of normal monkeys.6
The Adult group consisted of 13 monkeys, 10 adult feral animals and 2 juveniles and one infant born and reared in the group by their mothers. Additional information on living conditions and social behaviour of these monkeys may be found elsewhere.7
Six monkeys from each group were chosen as subjects. The two most dominant, two most subordinate, and two intermediate group members were selected on the basis of dominance tests.8 One of each of the dominant pairs, one subordinate from the juvenile group, and one intermediate from the adult group were males; all others were female.
The animals were housed in indoor-outdoor areas composed of approximately the following; 6.7 m2 of indoor caging with a grid floor and 14 m2 of outdoor floor space with solid floors; The roof of the outdoor pen area was covered with mesh open to the sky but half of the area had been subsequently covered with clear plastic sheeting which drained into the living area. The outdoor pens were 3.7 m high. Several ledges, bars and platforms provided a variety of outdoor elevated locations on which the monkeys could rest or travel. During the Woodchip conditions, 120 kg of woodchips were spread on the floor of each outside pen. No subsequent attention was paid to the woodchips. In the bare condition, the floor of the outdoor pens consisted only of bare, solid asbestos tiles which were cleaned daily in the morning by technicians using water and Tego.
Behaviours were recorded on to check sheets following two formats. The first, concerned with feeding behaviour, was designed to accommodate information on the rate of feeding of individual animals and on the number of animals on the ground at minute-long intervals. The second was concerned with 14 categories of social behaviour, 4 nonsocial behaviours, and the location of subjects. The behaviours recorded were as follows: huddle, groom, contact, play, proximity, sex, displace, threaten, chase, fight, be displaced, submit, flee, be attacked, walk, object manipulate, sit, and visual explore. The social categories were combined in such a way as to provide two primary measures: positive social interaction consisting of the first 6 behaviours, and negative social interaction of the second 8 behaviours. Walk and object manipulate, provided a composite index of non-social active behaviour, while sit and visual explore, represented a measure of nonsocial passive behaviour.
The following four conditions were of interest: Social behaviour in a bare pen with no food; social behaviour in a woodchip-covered pen with no food; social behaviour in a woodchip-covered pen with grain; feeding behaviour in a wood- chip pen with grain in the litter.
Four days of social behaviour testing were carried out in each of the two no-food conditions. Six monkeys were observed for a total of 20 minutes each, at 5 minutes per day giving a total of 120 minutes of observation per group for each condition. Data were collected in the form of modified frequencies using intervals of 15 seconds. If a behaviour occurred during this interval a score of one was recorded. These observations were carried out between 1300 and 1500 hours.
Feeding behaviours, testing in both grain conditions, consisted of four days of testing. On the first day 340 gm of dried maize (average diameter 8.3 mm) was scattered in the pen. If woodchips were present, the chips were then raked briefly to bury most of the grain. Each animal was observed for six one-minute long periods and the number of times any food items were transferred to the mouth recorded on a hand tally. At the end of every minute, the total number of animals from the group that were on the ground were noted. On the second day the same weight of pigeon food (diameter 7.6 mm) was similarly used. On the third day a mixed grain composed of 12% barley (diameter 4.7 mm), 27% milo (2.6), 16% white dari (3.7), 22% wheat (4.6), 19% sunflower (4.9), 2% maples (7.2), and 1% maize (8.3), by number was used for testing. The fourth day's grain was white millet (diameter 1.6 mm). As the grains differed in size, the number of individual items in a fixed weight was computed for the different grains. One hundred gram of the above four grains were estimated to contain 340, 370, 4 500 and 20 250 individual grains respectively.
An analysis of the cost of woodchips was undertaken as well as the smell of new condition. A new employee was asked to rate the two pens daily on a scale of from 0-4: none, slight, moderate, strong, or very strong smell respectively. This was done for four weeks with no woodchips in either pens and then for four weeks with woodchips but no other cleaning in one pen and normal cleaning but no deep litter floor covering in the other. At irregular intervals other individuals were asked for ratings of the two pens. Analyses of variance were used to assess the results. Alpha was set at 0.05. Analyses used three levels of dominance and two different pens as between subjects variables, and the presence or absence of chips as one of the repeated measures. One analysis tested time on the ground outside versus time spent above the ground outside; one analysis tested positive versus negative social behaviours; one tested nonsocial behaviours; one tested aggressive behaviour when given grain; one tested numbers of animals on the ground when given grain; and finally the rate of hand to mouth contacts, as a measure of the rate of grain consumption, was likewise tested. Data were also collected in the evening after feeding for the juvenile group, and these were tested against the afternoon observations.
RESULTS
In general the results showed that aggression was reduced in the presence of chips when grain was scattered; and after 6 weeks the chips smelled less than the bare pen after 24 hours.
When the four grains were offered aggressive behaviour was 4-6 times more frequent when the floor was bare as compared to with chips (F=27.0, df=l, 6, p=.002). This chips effect also interacted significantly with pen location (F=12.7, df=l, 6, p=.01). The frequency of aggression was 10 times greater for the pen containing the 15 juveniles and 1.8 times greater for the adult pen. There was no interaction with grain type/size although it appeared that the smaller grains were correlated with less aggression.
The analysis for positive and negative behaviours with no grain showed no significant effects related to the chips. There was a tendency, however, for positive behaviours to increase slightly and negative behaviours decreased by a factor of ten in the presence of the chips (F=3.7, df=l, 6, p=.10). The increase of positive behaviours was due to the subordinate monkeys, the decrease of negative behaviours was seen in all three ranks but was especially marked in the subordinate animals. There were no significant effects in the analysis of time on the ground when given grain.
There was a tendency to spend slightly more time on the ground with chips present (mean = 30.6 versus 27.2 sec.) than when bare, and the animals were longer on the bare ground with smaller, and therefore more numerous, grains.
The analysis of location of monkeys without grain present gave no significant effects of the presence of clean chips. In fact the animals spent slightly less time outside when chips were present and a slightly less percentage of that time on the floor (F=1.2, df=1 ,6, p=.31 ).
The analysis of nonsocial behaviours showed no significant effects of the presence or absence of chips. The amount of active nonsocial behaviours was increased threefold in the absence of chips, but this was not significant (F=1.1 , df=l, 6, p=.33).
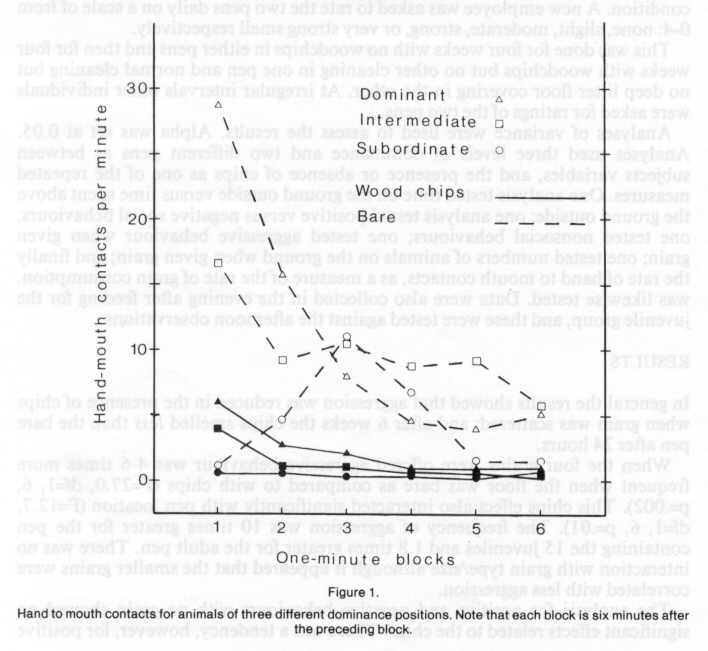
The analysis of hand to mouth contacts showed the most significant effects. There were significant differences in the following: Litter, grain, blocks, blocks x rank, blocks x rank x pen, litter x blocks, litter x blocks x rank, litter x blocks x pen, and litter x blocks x rank x pen (F=5.0, df=10, 30, p=.0003). Figure one illustrates these effects. Dominant animals feed faster at first and all animals feed faster in the bare pen. The difference in feeding rate is greatly reduced in the presence of chips. The interaction with pen is explained by the observation that the Juvenile " subordinate monkeys slid not show a peak of response during blocks 2, 3 and 4 in the bare condition. The more aggressive Juvenile animals inhibited foraging in the more subordinate members of the group. Also of interest was the significant grain effect; the smaller the grain size, the greater the number of hand to mouth contacts. The mixed grain and the millet showed over two and three times the number of hand-mouth contacts respectively as did maize, although millet was hardly eaten at all when it was raked into the wood chips, possibly because it could not be seen. When the juvenile pen was treated in the evening, there was no effect of chips. During the evening testing there was less positive behaviour and much more negative behaviour than during the afternoon (F=16.9, df=l, 3, p=.02). While the presence of chips reduced aggression by 2.5 times, this effect was not significant (F=.17, df=1, 3, p=.71).
Cleaning two pens every other day took one man 5 hours per week when no woodchips were present. Cleaning with woodchips took one man a total of 8.5 hours every four weeks. This included removal of the chips and cleaning the pen once in four weeks (3.5 hrs) and daily sweeping of the corridor when chips were tracked about by persons leaving the pens (1.5 hrs). The chips cost £2 per 40 kg bale.
The data on the amount of monkey smell from the pens shows that woodchips cleaned a minimum of every four weeks is less objectionable than a bare pen cleaned every other day. The average rating for the bare pen was 1.02, slight; the average rating for the pen with chips was 0.58, between none and slight. The bare pen received five strong and five moderate ratings whereas the highest rating of the chip pen was a single moderate rating. Two pens were left six weeks and still naive raters not working with animals of any kind could only detect the smell of the wood chips and no smell of faeces or urine.
DISCUSSION
This brief study has shown that the presence of woodchips on the floor of a living pen has no detrimental effects and has several beneficial ones. These are the reduction of fighting, less smell, improved appearances, and more equal feeding rates. We have also noticed an improved cleanliness of the monkeys' coats, cleaner windows, and some noise attenuation. During the study we were careful to use chips with no food in it. After the study was completed and when grain was given at twice- to thrice-weekly intervals, we have noticed that animals spend a considerable time foraging through the chips, in fact 30 times as much as when chips were first introduced and had no food in it, i.e., the behaviour analysis in this report. This probably contributes towards decreased fighting in the animals.
The experiment took place during a wet summer, and half the chips were often wet from rain. The monkeys showed less preference for this area than for the dry area. We have concluded that in a stable, disease free colony, living under cover, woodchips are a preferable floor covering over a bare floor. Furthermore, it is an easily reversible flooring condition.
References
1. N.A.S. (1968). Nonhuman primates. National Academy of Sciences, Publication 1677, Washington.
2. DHEW (1972). Guide for the care and use of laboratory animals. Department of Health Education and Welfare, Publication number (NIH) 73-33, Washington.
3. Whitney, R. A., Johnson, D. J. and Cole, W. J. (1973). Laboratory primate handbook, Academic Press, London.
4. Kingston, W. R. (1969). Marmosets and tamarins. Laboratory Animal Handbook, 4, 243-250.
5. Chamove, A. S. (1973). Varying infant rhesus social housing. Journal of the Institute of Animal Technicians, 24, 5-15.
6. Chamove, A. S. (1975). Self-feeding infant macaques. The Simian, 18, 3-5.
7. Anderson, J. R. and Chamove, A. S. (in press). Contact and separation in adult monkeys, South African Journal of Psychology.
8. Chamove, A. S. and Bowman, R. E. (1978). Rhesus plasma cortisol response at four dominance positions. Aggressive Behaviour, 4, 43-55.
Reproduced with permission of the Institute of Animal Technology.