Viktor Reinhardt
Wisconsin Regional Primate Research Center,
University of Wisconsin, Madison
Received for publication March 7, 1991; revision accepted October 8, 1991.
The sequence in which 14 laboratory rhesus macaques left their home enclosure during a routine catching procedure was recorded on 30 occasions during 6 weeks. The animals were trained to voluntarily exit one by one and enter a transport cage for weighing and/or treatment.
Mean weekly exit orders cross-correlated significantly, and individuals retained their exit positions with remarkable consistency throughout the study period. An animal's position did not depend on its sex, age, or dominance rank. The oldest female never failed to enter the transport cage as number 1. When she was prevented from taking the lead, the group was hesitant but eventually proceeded in the usual order. Food was a strong, yet not absolute, incentive for the monkeys to cooperate. If they could not expect a reward, they took more time before proceeding, but did so in the habitual order.
The predictability of the exit sequence has practical managerial value because it makes it possible for a single person to quickly catch a selected group member when necessary.
Key words: macaques, management, spatial order
INTRODUCTION
In zoos and laboratories, individual animals from groups of macaques must be caught regularly for weighing, tuberculosis testing, health control, pregnancy monitoring, blood sampling, stool/urine sampling, medical treatment, etc. The capture procedure is usually distressing for the monkeys and for the personnel. The animals often show signs of anxiety and distress (e.g., grimacing, crouching, screaming, temporary diarrhea), and their reluctance to being caught is often countered by using forceful means (e.g., yelling at the animals, chasing the animals with sticks into transport boxes, chasing and catching individual animals with special nets).
A simple training technique has recently been described for ensuring the cooperation of group-housed rhesus macaques during routine catching of all or individual members of a group [Reinhardt, 1990]. The training was implemented in an attempt to minimize stress reactions of the animals and to increase the safety of the personnel.
The present investigation examines the sequence in which trained rhesus macaques leave their familiar enclosure in order to be caught, as well as the consistency over time and the structural elements of this sequence. To the knowledge of the author, voluntary progression orders have not been described in any macaque species in feral or captive groups. The only primates studied have been wild baboons, but given the constraints of field observations, no invariant spatial movement patterns have been described for individual animals [Hall and DeVore, 1965; Kummer, 1968; Altmann and Altmann, 1970; Rhine, 1975; Rhine and Westlund, 1981; Giacoma and Messeri, 1981; Rhine et al., 1985; Rhine and Tilson, 1987].
MATERIALS AND METHODS
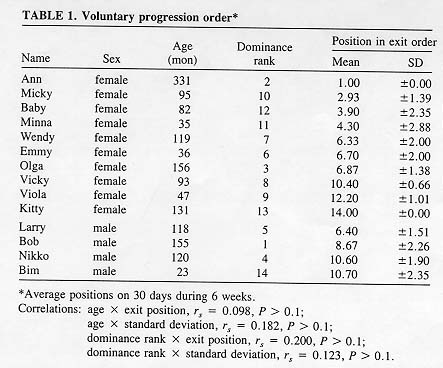
Fourteen healthy rhesus macaques (Macaca mulatta) were the subjects of this study, including four males and ten females, two of which had nursing infants. The animals ranged in age from 2 to 28 years. Their dominance-subordination relationships were known and verified during daily check-ups. Between two animals, the subordinate partner would consistently yield and/or grimace. A subject's dominance rank was determined by the number of its subordinate partners. Rank 1indicated the highest, rank 14 the lowest position in the group's dominance order. Males and females did not differ significantly in age (U = 20; Table 1) or in dominance rank (U = 14; Table 1).
The 14 rhesus macaques had lived together as a compatible group for several years in an indoor pen 5.7 m deep, 2.5 m wide, and 2.1 m high. The pen was subdivided by a wire mesh wall into a large home area (10.6 m2 ) and a small holding compartment (3.5 m2). Commercial dry food was provided at 0730, supplemented with fruit at 1500. Water was available ad libitum. Room temperature was maintained at 20-22ºC, with a 12-hr light/dark cycle.
The troop had been trained I year prior to this study to enter the holding compartment on command and voluntarily exit one at a time through a chute into a transport cage [Reinhardt, 1990]. The training was based on positive reinforcement, and cooperative behavior was rewarded with fruit [Reinhardt, 1990]. The animals were accustomed to being caught at least once every other month for weighing or individual treatment.
The sequence in which the monkeys left the holding compartment and entered the transport cage, delays in the exit order (an animal entering the transport cage more than 30 sec after the door was opened), and total time required to catch all 14 subjects were recorded under the following circumstances:
Weeks 1-4: The group was confined in the holding compartment at 1500 and fruit was distributed in the home area. All group members were caught one by one, briefly kept in the transport cage, and then released back into the home area. This procedure was carried out each day from Monday through Friday for 4 consecutive weeks.
Weeks 5-6: The animals received their daily fruit ration I hr before the catching procedure. Again, the group was confined in the holding compartment at 1500 and the animals caught, briefly kept in the transport cage, and released back into the home area. This procedure was carried out again each day from Monday through Friday for 2 consecutive weeks.
Week 7: On Monday, the leading animal was caught and placed in a transport cage outside the holding compartment. An attempt was then made to catch the rest of the group as usual, with fruit waiting for them in the home area.
Mean position in the sequence in which the animals left the holding compartment was calculated for each individual for weeks 1-6. The consistency of individual animals' positions in the progression order was evaluated by computing their standard deviations.
Statistical analysis was done using the Mann Whitney U test, Student's t test and Spearman's rank test [Siegel, 1956]. The level of significance was set at P < 0.05.
RESULTS
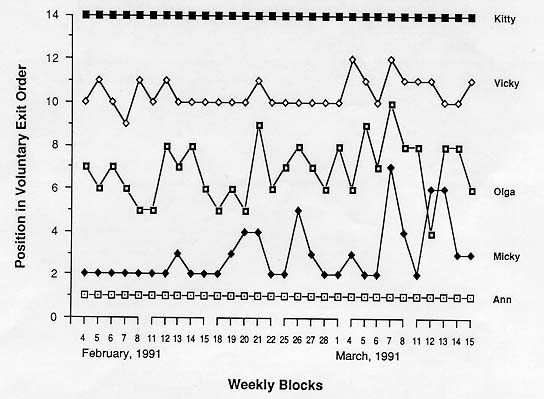
The 14 rhesus macaques left the holding compartment of their pen and voluntarily entered the transport cage one by one in a predictable order. Figure I depicts examples of five different animals. The mean weekly exit orders of weeks 1-6 cross-correlated significantly (rs always > 0.100).
Standard deviations of mean exit positions during weeks 5 and 6 did not differ significantly from those during weeks 1-4 (mean5 = 1. 8 ± 1.1 vs. mean6 = 1. 2 ± 0.6; t = 1.834). During weeks 5 and 6, however, the animals were more hesitant to exit and daily progressions were delayed significantly more often than during weeks 1-4 (mean5 = 4.6 ± 1.6 times vs. mean6 = 2.6 ± 1.6 times; t = 3.515). As a consequence, significantly more time was required to catch all 14 animals during weeks 5 and 6 than during weeks 1-4 (mean5= 26.2 ± 13.8 min vs. mean6 = 9.8 ± 2.1 min; t = 3.741).
Mean exit positions and their standard deviations during weeks 1-6 were correlated neither with age nor with dominance rank (Table 1). Old or high-ranking group members did not leave the holding compartment earlier than young or low-ranking group members, nor did they retain a special position in the exit order with greater consistency.
Exit positions of males did not differ significantly from those of females (U = 12; Table 1). One of the two mothers with nursing infants ranked last in the progression order (Kitty, Table 1), whereas the other was one of the first animals in the progression order (Wendy, Table 1).
Female Ann was the first in the exit order on all 30 occasions during weeks 1-6 (Fig. 1). She never failed to take the lead within 30 sec after the author opened the exit door to the transport cage. Ann was the oldest but not the most dominant animal (Table 1). On Monday of week 7, Ann was not released into the home area but placed outside the holding compartment. Initially no other member of the group made an attempt to leave. After a delay of 8 min, Nikko, who usually was one of the last animals to exit (Table 1), left the holding compartment to be caught and released back into the home area; he was promptly followed by the rest of the group in the habitual sequence.
DISCUSSION
The rhesus macaques in this study were trained to cooperate when being caught one by one. Despite the marked regularity of the exit order, there was no apparent structuring element. The order was dependent neither on age nor on dominance rank, and the consistency with which animals retained their positions was also related neither to age nor to dominance rank. This contrasts with the situation in which rhesus macaques are routinely forced to leave their pen. Under such a circumstance, the progression order is also stable, but it is determined by age, with old animals leaving before young ones and retaining their positions with greater consistency [Reinhardt and Reinhardt, 1987]. It has been suggested that when being routinely forced to leave the pen, individuals learn with age to overcome their anxiety with the prospect of quickly returning to the home area; hence they proceed in accordance with age rather than in accordance with dominance [Reinhardt and Reinhardt, 1987]. When being trained to cooperate, the animals apparently experience negligible or even no anxiety, and no age-dependent learning influences their decision to proceed. The very same situation has been described in cattle, which also show stable progression orders under forced and voluntary conditions. As with rhesus macaques, age determines an individual's position in the fear-induced but not in the spontaneous progression order in cattle [Reinhardt, 1983].
A food reward as well as the leader of the group were strong, yet not absolute, incentives for the rhesus macaques to cooperate during the catching procedure. If they could not expect a reward or if the leader was prevented from proceeding, individual animals were more hesitant but left the holding compartment in the usual order. This underscores that the animals' training-conditioned behavior was relatively fixed.
The reliability of the exit order has desirable managerial applications because it allows quick and safe catching of a specific individual if need arises (e.g., disease, injury, pregnancy check): all animals that have lower positions in the exit order than the selected subject are released from the holding compartment directly into the home area. The caretaker then closes the chute exit, opens the entrance to the transport cage, and catches the selected monkey as it follows the progression order. The predictable exit position of the selected animal makes it unnecessary to dyemark it beforehand. Catching a specific subject does not require more than 5 min and can be achieved at any time by a single handler. It is tempting to speculate that other group-living primates such as baboons, lion-tailed macaques, stump-tailed macaques, Celebes macaques, and capuchin monkeys can be trained as readily as rhesus macaques to cooperate predictably during the catching procedure, thereby avoiding unnecessary turmoil and the risks (e.g., aggression, abortion) associated with it.
ACKNOWLEDGMENTS
I am thankful to my wife Annie and to three anonymous reviewers for providing very helpful comments on this manuscript, and to Mr. John Wolf for editing it. This project was supported by NIH grant RR-00167 to the Wisconsin Regional Primate Research Center.
REFERENCES
Altmann, S.A.; Altmann, J. Baboon Ecology. Chicago, University of Chicago Press, 1970.
Giacoma, C.; Messeri, P. Spatial and social relationships among male hamadryas baboons. Monitore Zoologico15:314, 1981.
Kummer, H. Two variations in the social organization of baboons. Pp. 293-312 in Primates. P. Jay, ed. New York, Holt, Rinehart & Winston, 1968.
Hall, K.R.L.; DeVore, I. Baboon social behavior. Pp. 53 -110 in Primate Behavior: Field Studies of Monkeys and Apes. I. DeVore, ed. New York, Holt, Rinehart & Winston, 1965.
Reinhardt, V. Movement orders and leadership in a semi-wild cattle herd. Behaviour 83:251264, 1983.
Reinhardt, V.; Reinhardt, A. Prompted progression order in a troop of captive rhesus monkeys. Folia Primatologica 48:121-124, 1987.
Reinhardt, V. Avoiding undue stress: Catching individual animals in groups of rhesus monkeys. Lab Animal 19:52-53,1990.
Rhine, R.J. The order of movement of yellow baboons (Papio cynocephalus). Folia Primatologica 23:72-104, 1975.
Rhine, R.J.; Westlund, B.F. Adult male positioning in baboon progressions: Order and chaos revisited. Folia Primatologica 35:77-116, 1981.
Rhine, R.J.; Bloland, P.; Lodwick, L. Progressions of adult male chacma baboons (Papio ursinus) in the Moremi Wildlife Reserve. International Journal of Primatology 6:115-122, 1985.
Rhine R.J.; Tilson, R. Reactions to fear as a proximate factor in the sociospatial organization of baboon progressions. American Journal of Primatology 13:119-128, 1987.
Siegel, S. Nonparametric Statistic for the Behavioral Sciences. New York, McGraw-Hill, 1956.
This article originally appeared in Zoo Biology 11, 61-66 (1992).
Reprinted with permission of the Editor.