Linda Brent, D. Rick Lee, Jorg W. Eichberg
Department of Virology and Immunology,
Southwest Foundation for Biomedical Research,
San Antonio, TX 78284 Antonio, TX 78228-0147
Accepted for publication March 16, 1990.
Key words: area use - environmental enrichment - species-typical behavior - well-being
A large, three-part playground for captive chimpanzees was constructed and evaluated in terms of area use and behavior changes. Comparative behavioral samples were obtained on 38 subjects in the existing indoor-outdoor run and in the enclosure. The chimpanzees used the inside run, connective chute, concrete slab, and grass areas most. Activity and environmental manipulation increased in the enclosure while abnormal and self-directed behaviors decreased .
Introduction
The recent interest in animal welfare has focused much attention on the environment of captive animals, especially the chimpanzee. Impoverished captive environments have been associated with increased abnormal and maladaptive behaviors, and decreased reproductive competence and species-typical behaviors [6,12,15]. Improvement of the environment, or environmental enrichment, has been studied in nonhuman primates as a means to alleviate these problems.
The factors affecting an animal's habitat, including the social environment, available area, and variety of furnishings [9,32], have been the subjects of various environmental enrichment programs. For many nonhuman primates, including chimpanzees, social stimulation is required for adequate behavioral development [10,28]. The addition of social partners for environmental enrichment was studied with macaques and provided an excellent source of stimulation [251].
Simply increasing cage size as a means of enrichment has met with inconclusive results. Increased cage size has been found to have no effect on chimpanzees and has even increased abnormal behavior of rhesus monkeys [2,31]. Descriptions of chimpanzee groups moved to large, outdoor habitats have included both positive and negative behavioral or physical consequences [16,241].
Providing manipulable objects for captive nonhuman primates does not require changes in social groups or physical housing practices, and can also be cost effective. Rubber toys, trees, and artificial termite mounds have had positive effects on primate behavior [7,18,22]. The enrichment device must also be specific to the needs of the individual and the species. Hard dog toys were rarely used by a group of captive cynomolgus monkeys, and a chimpanzee feeding puzzle was associated with individually differing behavior changes [4,17].
From the enrichment studies, it is evident that many environmental factors influence the well-being of the captive primate. For example, the activity of apes was affected by both the number of animals and types of objects in the enclosure [31]. Providing a variety of environmental enrichment devices (including social, spatial, and complexity factors) takes into account the species' total environment and individual differences. Several investigators have quantitatively studied the effects of this approach on apes, with generally positive results [5,81].
Clearly, more research on a multifaceted enrichment approach is necessary to substantiate the advantages for nonhuman primates. To fully utilize captive animal resources, biomedical research facilities can also accommodate behavioral research. The construction of an enrichment facility at the Southwest Foundation for Biomedical Research, San Antonio, TX, provided the means to study the effect of a combination of increased space and complexity for socially housed chimpanzees. Use of area and behavioral changes when the animals were in the enclosure were studied to determine if this avenue of enrichment was beneficial for the subjects. An empirical basis for evaluation, as well as consideration of cost and management practices, is necessary when recommending such an enrichment enclosure for improvement of the captive nonhuman primate environment.
Methods
Subjects
Thirty-eight chimpanzees (Pan troglodytes), housed in social groups of three to nine individuals, were the subjects of the study. Ten males, 28 females, and an additional eight infants were housed in the nine indoor-outdoor runs of the breeding facility. The adults ranged in age from seven to 35 years, with an average age of 21 years, at the beginning of the study. All animals had been housed in this facility or a similar building for several years. The chimpanzees were fed a diet consisting of fruit, vegetables, and commercially available monkey chow.
After the completion of the three enrichment enclosures. or playgrounds as they became known, the nine chimpanzee groups were rotated into the playground every third week, after spending two weeks in the indoor-outdoor run only. While occupying the playground, each group still had continuous access to the indoor run, where they were fed, to facilitate regular observation and capture if necessary.
The enrichment enclosure
The 9,000 sq. ft. (2,744 m2) facility consisted of three enclosures with walls of heavy chain link panels and a bar roof [33] connected to the existing chimpanzee breeding facility at the Southwest Foundation for Biomedical Research, San Antonio, TX. Each measured 40 ft. wide X 75 ft. long (12.2 m wide X 22.9 m long). The ground covering was grass with a 10 ft. (3 m) concrete slab next to the indoor-outdoor runs of the breeding facility. A variety of equipment, constructed mainly of pipe, rope, tires, culverts, and chain, was added to each enrichment area. The items provided shade and visual barriers, and allowed use of both vertical and honizontal dimensions of the playground. Access to the enclosure from the existing runs was provided by an overhead chute that could be closed at either end. Water was available in each enclosure.
Data collection and analysis
Data collecting sessions consisted of four to ten min focal animal samples in which durations of all occurrences of specified behaviors were recorded on a portable computer (Tandy 1400 LT) for a single subject [1]. Data were recorded between 0800, 1100 and 1400-1600 weekdays between April and September 1989. Since use of area may vary with novelty [21], a ten-week pilot study was completed initially to familiarize the subjects with the playground and the observer. During this time each group had access to the playground for at least three weeks. Following this period, at least 60 min of observation were completed for each animal while in the playground, and another 60 min while in the indoor-outdoor run. A total of 75 hr of observation were completed.
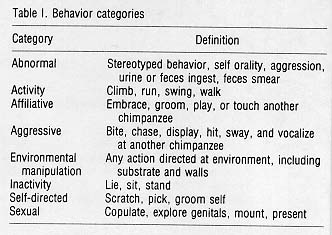
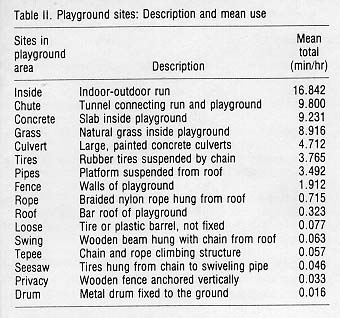
For each sample, the context, behavior, recipient, site, and duration were recorded. Forty-seven behaviors were defined and grouped into eight context categories (see Table 1). Thirty-five playground sites were defined, recorded, and placed in 16 categories (see Table 2). All data were transformed into min/hr/subject, after subtracting the out-of-sight total. The analysis included determining the use of areas in the playground and a comparison of behaviors between the indoor-outdoor run and the playground. Four subjects were deleted from the behavioral analysis due to changes in group composition for management reasons. Data were analyzed with the SAS statistical package and a significance level of p < 0.05 was defined.
Results
The opening of the enrichment facility was on a cool day in April 1989. Staff members gathered outside to await the emergence of the first group of chimpanzees. Although all animals were initially hesitant, they usually ran through the chute to the playground within 30 min of the opening. The soft grass was unfamiliar to chimpanzees reared on concrete and wire, and several preferred to climb along the fence and roof during the first week. However, 65% of the chimpanzees walked on the grass the first day. Initially, the comer perches, culverts, pipe platform, and loose items were used most often. The swinging tires, ropes, and moveable climbing structures were the last areas to be explored. Tree branches with leaves were periodically placed into the playgrounds. The chimps used the branches, as well as the grass, as browse material and for building nests. Additional items, such as unshelled nuts and popcorn, were also thrown into the playground from above to stimulate foraging behavior. After several weeks in the playground, most animals freely walked in the grass, utilized all of the playground equipment to various degrees, and were habituated to the presence of an observer. At this point, the reported data collection began.
Use of playground areas
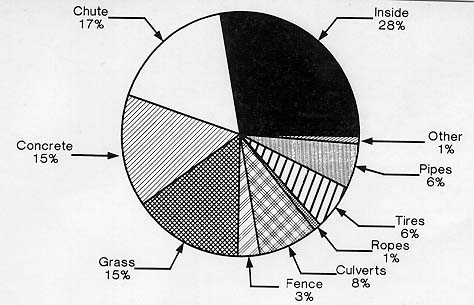
Sixteen sites in the playground were analyzed for each group and analysis of variance showed significant differences in means (F(15,128) = 15.32, p < .001) (see Table 2). Further analysis with the Ryan-Einot-Gabriel-Welsch Multiple Range test was completed to separate means [11]. The inside, chute, concrete, and grass area were used significantly more than the other areas (df = 120) (see Fig. 1).
Similar analysis of major areas was completed by combining single areas and dividing by the total number of areas to obtain a per unit measure. The means for the comers, edges, and middle of the playground were 2.658, 1.337, and 0.253 respectively. The comer mean was significantly greater than the middle (F(2,24) = 7.45, p < .005). The use of edges was not significantly different from comer or middle use. No significant difference was found between the use of areas on or above the ground in the playground.
Male and female differences were significant only for the concrete mean (F(1,37) = 11.80, p < .002) and the loose objects mean (F(1,37) = 6.83, p < .0 13). In both cases, the males' use was greater than the females'. Age was negatively correlated with inside (r = - .4015, p > .0 13) and fence areas (r = - .3685, p > .023).
Behaviors
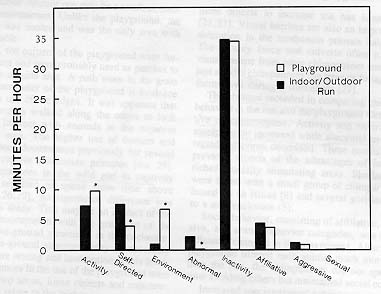
The behaviors were grouped into eight categories and each analyzed with repeated measures ANOVA procedures by social group. The comparison of the behavior categories between the indoor-outdoor run and playground provided several significant results (Fig. 2). Activity was significantly increased from the run to the playground (F(1,25) = 9.363, p < .005). Environmental manipulation was significantly increased in the playground (F(1,25) = 23.330, p < .001). Separating the grass manipulation category from the analysis still provided a significant environmental increase (F(1,25) = 9.308, p < .001) as well as a significant grass manipulation increase (F(1,25) = 14.856, p < .001) from the run to the playground. Self-directed (F(1,25) = 43.455, p < .001) and abnormal behaviors (F(1,25) = 16.860, p < .001) were both significantly decreased from the run to the playground. Inactivity, and affiliative, aggressive, and sexual behaviors in the playground were not significantly different from those in the run.
Grouping effects were significant for activity (F(8,25) = 3.2131, p < .012), self (F(8.25) = 3.520, p < .007) and abnormal behavior categories (F(8,25) = 2.4 10, p < .044). To determine if other factors may have affected the groups, a comparison with age and sex was completed. Age was significant for both activity and abnormal behaviors (F(1,30) = 13.578, p < .001, (F(1,30) = 4.560, p < .041, respectively). An analysis of behaviors by sex showed the aggressive category mean significantly greater for males than females (F(1,32) = 23.64, p < .001); however, no other comparisons were significant.
BEHAVIOR CATEGORIES
*Signifcant difference (<0.05).
Discussion
The results indicated that captive chimpanzees in an enrichment enclosure use areas of the enclosure differentially. The use of certain locations has been related to better visibility, potential escape routes, food and water resources, and secure sleeping sites [30]. The areas with the most frequent mean use were the inside, chute, concrete, and grass areas. An explanation may lie in the relationship between the chimpanzees' predominant behaviors and the characteristics of the preferred areas. Since over half of their time was spent resting, the level, non-moveable sites may have been preferred for this purpose. The large amount of time spent inside the existing indoor-outdoor run may be a result of comfort and convenience. Unlike the playground, the inside run was cooled and was the only area with food available.
Overall, the comers of the playground were frequented most and were probably used as perches to observe the outside area. A path worn in the grass along the perimeter of the playground is evidence of the high use of the edges. It was apparent that the chimpanzees walked along the edges to look outside or interact with animals in the adjacent playground area. The higher use of comers and edges has been documented previously for several species of captive nonhuman primates [see 29]. While chimpanzees in the wild and in captivity have been reported to spend more time above ground [13,26,29], no significant difference was found in this study. This may be an artifact of the subjects' preoccupation with the grass, or of the type of above-ground structures in the enclosure. Larger above-ground structures may be necessary to induce more resting and locomotion at this level.
Sex differences in the use of the playground were evident for two areas, loose objects and concrete, and probably relate to the higher aggressive behavior level recorded for males. The loose tire and barrel were often thrown or rolled during displays. This parallels the behavior of wild chimpanzees, who regularly drag, throw, and shake objects to accentuate displays [141]. In addition, male chimpanzees displayed and vocalized from the concrete area to other chimpanzee groups in the existing runs. No injuries have occurred in the playground.
Younger chimpanzees were likely to use the fence and inside run more often than older animals. While use of the fence may be due to greater agility of younger chimpanzees, the inside use contrasts with reports of higher levels of exploratory behavior in young chimpanzees [14]. However, factors particular to this study may have had an effect. The younger chimpanzees composed a social group with less stability than the groups of adults. Perhaps the increased exposure to neighboring chimpanzees in the playground was more threatening for younger animals without clear-cut dominance structures.
The chimpanzees' use of playground areas provides guidelines for constructing an enrichment enclosure. The quality of the area should be considered as much as the quantity of space available [8,19]. Inexpensive, durable materials, such as tires, culverts, and chains, can be used in a variety of ways to add complexity to the environment. The placement of such materials can accentuate use of vertical space, an important consideration for ape habitats [201]. Loose objects, food items, and foraging material can be easily added and removed from the enclosure. The importance of varying enrichment objects to increase use has been reported [21,23]. Visual barriers are also an important consideration in the nonhuman primate habitat [30]. The privacy fence and culverts often provided a visual escape from other chimpanzees and visitors, just as wild chimpanzees use trees or foliage to hide themselves during disturbances [27].
The changes recorded in comparing chimpanzee behavior in the run and the playground were indicative of improvement. Activity and environmental manipulation increased while abnormal and self directed behaviors decreased. These results confirm previous reports of the advantages of large, enriched, socially stimulating areas. Similar results were found with a small group of chimpanzees released on an island [8] and several gorillas moved to a new enclosure [5].
Social behavior, consisting of affiliative, aggressive, and sexual behavior categories, was not significantly changed in the playground. Although the playground offered each animal much more space than the run, the chimpanzees did not spend the time avoiding others but maintained social contacts.
Increased species-typical behavior patterns have been used as a way to determine increased well-being [18]. Foraging, nest building, using objects in displays, and increased locomotion patterns were examples of the behaviors evident in the playground area. Although this environment was not constructed to duplicate the natural environment of wild chimpanzees, it was possible to facilitate the expression of species-typical behaviors in a stimulating man-made enclosure.
The combination of enrichment strategies present in this outdoor facility has proven to be beneficial for captive chimpanzees. Providing social stimulation, large areas for exploration and locomotion, and various devices within the enclosure have resulted in behaviors more similar to those of wild chimpanzees. The reported use of area and accompanying behavioral changes can be used as evidence to support and facilitate the construction of similar environmental enrichment projects for captive chimpanzees.
Acknowledgments
We wish to thank Humberto Barrera for his assistance in designing and constructing the playgrounds.
References
1. ALTMANN J: Observational study of behavior: sampling methods. Behaviour 49:227-267, 1974.
2. BAYNE KAL, MCCULLY C: The effect of cage size on the behavior of individually housed rhesus monkeys. Lab Anim 18(7):25-28, 1989.
3. BERKSON G, MASON WA, SAXON SV: Situation and stimulus effects on stereotyped behaviors of chimpanzees. J Comp Physiol Psychol 56:786-792, 1963.
4. BLOOMSTRAND M, RIDDLE K, ALFORD P, MAPLE TL: Objective evaluation of a behavioral enrichment device for captive chimpanzees (Pan troglodytes). Zoo Biol 5:293-300, 1986.
5. BOWEN RA: The behaviour of three hand-reared lowland gorillas, Gorilla g. gorilla with emphasis on the response to a change in accommodation. Dodo, J Jersey Wildl Preserv Trust 17:63-79, 1980.
6. BRENT L, LEE DR, EICHBERG JW: The effects of single caging on chimpanzee behavior. Lab Anim Sci 39(4): 345-346,1989.
7. BRENT L, LEE DR, EICHBERG JW: Evaluation of two environmental enrichment devices for singly caged chimpanzees. Am J Primatol Suppl 1:65-70, 1989.
8. CLARKE AS, JUNO CJ, MAPLE TL: Behavioral effects of a change in the physical environment: A pilot study of captive chimpanzees. Zoo Biol 1:371-380, 1982.
9. COE JC: Naturalizing habitats for captive primates. Zoo Biol Suppl 1: 117-125, 1989.
10. DAVENPORT RK, ROGERS CM: Differential rearing of the chimpanzee: A project survey. Behaviour 3:337-360, 1970.
11. EINOT 1, GABRIEL KR: A study of the powers of several methods of multiple comparisons. J Am Stat Assoc 70:351. 1975.
12. ERWIN J, DEM R: Strangers in a strange land: Abnormal behaviors or abnormal environments? In Erwin J. Maple TL. Mitchell G (eds): "Captivity and Behavior: Primates in Breeding Colonies, Laboratories. and Zoos. " New York: Van Nostrand Reinhold Co., 1979, 1-28.
13. GOODALL J: Chimpanzees of the Gombe Stream Reserve. In DeVore I (ed): "Primate Behavior: Field Studies of Monkeys and Apes. New York: Holt Rinehart and Winston. 1965, 425-473.
14. GOODALL J: "The Chimpanzees of Gombe: Patterns of Behavior. " Cambridge: The Belknap Press of Harvard University Press, 1986.
15. HOOFF JARAM VAN: The Arnhem Zoo chimpanzee consortium: an attempt to create an ecologically and socially acceptable habitat. Int Zoo Yearbook 13:195-203, 1973.
16. KOLLAR EJ, EDGERTON RB, BECKWITH WC: An evaluation of the behavior of the ARL colony chimpanzees. Arch Gen Psychiat 19:580-594, 1968.
17. LINE SW: Environmental enrichment for laboratory primates. J Am Vet Med Assoc 190:854-859, 1987.
18. MAKI S, BLOOMSMITH MA: Uprooted trees facilitate the psychological well-being of captive chimpanzees. Zoo Biol 8:79-87, 1989.
19. MALLINSON JC: Cage furnishings and environments for primates with special reference to marmosets and anthropoid apes. Zoo Fed News 33(Suppl 12):1-8, 1982.
20. MAPLE TL: Great apes in captivity: The good, the bad, and the ugly. In Erwin J, Maple TL, Mitchell G (eds): "Captivity and Behavior-Primates in Breeding Colonies, Laboratories, and Zoos." New York: Van Nostrand Reinhold Co., 1979, 239-272.
21. MENZEL EW JR: Group behavior in young chimpanzees: responsiveness to cumulative novel changes in a large outdoor enclosure. J Comp Physiol Psychol 74:46-51, 1971.
22. NASH VJ: Tool use by captive chimpanzees at an artificial termite mound. Zoo Biol 1:211-221, 1982.
23. PAQUETTE D, PRESCOTT J: Use of novel objects to enhance environments of captive chimpanzees. Zoo Biol 7:15-23, 1988.
24. PFEIFFER AJ, KOEBNER LJ: The resocialization of single-caged chimpanzees and the establishment of an island colony. J Med Primatol 7:70-81, 1978.
25. REINHARDT V: Behavioral responses of unrelated adult male rhesus monkeys familiarized and paired for the purpose of environmental enrichment. Am J Primatol 17:243-248, 1989.
26. REYNOLDS V, REYNOLDS F: Chimpanzees of the Budongo Forest. In DeVore I (ed): "Primate Behavior. Field Studies of Monkeys and Apes." New York: Holt. Rinehart and Winston, 1965, 368-424.
27. REYNOLDS V, REYNOLDS F: The natural environment and behaviour of chimpanzees Pan troglodytes schweinfurthi and suggestions for their care in zoos. Int Zoo Yearbk 5:141-144, 1965.
28. SUOMI SJ, COLLINS ML, HARLOw HF: Effects of permanent separation from mother on infant monkeys. Dev Psychol 9:376-384, 1973.
29. TRAYLOR-HOLZER K, FRITZ P: Utilization of space by adult and juvenile groups of captive chimpanzees (Pan troglodytes). Zoo Biol 4:115-127. 1985.
30. WILSON CC: Spatial factors and the behavior of nonhuman primates. Folia Primatol 18:256-275, 1972.
31. WILSON SF: Environmental influences on the activity of captive apes. Zoo Biol 1:201-209. 1982.
32. WOOLVERTON WL, ATOR NA. BEARDSLEY PM. CARROLL ME: Effects of environmental conditions on the psychological well-being of primates: A review of the literature. Life Sci 44:901-917, 1989.
33. EICHBERG JW, LEE DR, BUTLER TM, KELLEY J, BRENT L: Construction of playgrounds for chimpanzees in biomedical research. J Med Primatol 20:12-16, 1991.
This article originally appeared in the Journal of Medical Primatology 20, 29-34 (1991).
Reprinted with permission of the Editor.