E Ludes-Fraulob1 and J R Anderson2
1Centre de Primatologie, Université Louis Pasteur, 67207 Niederhausbergen, France
2Department of Psychology, University of Stirling, Stirling FK9 4LA, UK
Final Acceptance: 27 August 1998
Abstract
A captive group of white-faced capuchins, Cebus capucinus, was presented with four deep litters in simultaneous choice (or preference) tests. A floor covering of ground corn cob, woodchips, wood wool or peat was presented once in each quarter of the group's indoor floor-area for 14 consecutive days, and the layout of the litters was rotated after each such period. The monkeys were observed on 10 days in each period to determine the occurrence of locomotion, foraging, play, and social contact on each of the litters. The ground corn cob was clearly the least attractive floor covering for the monkeys, while peat and wood wool proved to be the most attractive. Most instances of social contact occurred on the peat, due to the occurrence of communal peat-bathing, while wood wool afforded the most play. The provision of different litter types in different areas of the indoor enclosure is a simple means of promoting a greater range of natural activities in captive primates, and probably also in other animals.
Keywords: animal welfare, behaviour, foraging, litter, play, preferences, social contact
Introduction
Over the last 20 years there have been several reports of positive behavioural changes in captive non- human primates following the provision of deep litter, i.e. a floor covering, usually of organic origin, which promotes one or more of a range of activities including locomotion, foraging, and play. In general, the changes reported involved some shift in the animals' behavioural profiles nearer to those which might be observed in their wild counterparts, so the provision of litter is seen as environmental enrichment (Chamove & Anderson 1988; 1989; Poole 1988; Reinhardt & Roberts 1997).
The first study of the effects of providing a group of laboratory macaques with woodchip litter (Chamove & Anderson 1979) found fewer aggressive interactions when small food items were scattered onto the litter compared with the bare floor of the pen. Foraging time was increased in the presence of the litter, and reduced dominance -related competition was expressed through smaller differences in feeding rates. Subsequent studies increased the range of litter types and primate species assessed, and found further effects including increased overall activity (Chamove et al 1982; Boccia 1989; Byrne & Suomi 1991; Brent 1992; Ludes & Anderson 1996), reductions in abnormal behaviours (Anderson & Chamove 1984; Rosenblum & Smiley 1984), and a rebound in foraging activity after a period of litter deprivation, even when the litter contained no food items (Anderson & Chamove 1984). This latter effect suggests that litter-facilitated foraging reflects a behavioural drive which might be inadequately met in environments which are devoid of suitably stimulating foraging substrates.
Surprisingly, and in spite of the accumulation of evidence for beneficial effects of deep litter for captive primates, evaluations have neglected a powerful method used in other applied animal behavioural domains, namely simultaneous preference (or choice) tests. Direct measurement of preferences is being used increasingly to provide insights into species-relevant features in the captive environment; for example in laboratory rodents (e.g. Mulder 1975; Manser et al 1994; van de Weerd et al 1997), farm mammals (Natzke et al 1982; Fraser 1985) and poultry (Hughes & Black 1973; Dawkins 1977; Petherick & Duncan 1989). Although factors such as artificial selection, motivational state and previous experience with particular environments need to be considered when employing choice tests (Duncan 1978), this approach can provide a reliable means of assessing animals' relative preferences among particular environments; and as such it is a useful starting point for assessing an animal's evaluation of its environment (Dawkins 1983; 1990; Fraser et al 1993; Mench 1994).
Anderson and Chamove (1984) stated briefly that a captive group of stump-tailed macaques, Macaca arctoides, played more in, and showed more manipulation of, a wood wool litter as compared with a simultaneously present woodchip litter over 2-day periods. However, the present study is the first to apply a simultaneous choice method systematically, to gauge preferences in non-human primates when presented with different types of litter over relatively long periods. As the capuchin monkeys in this study had previously been studied for reactions to each litter using the more common method of baseline versus a single litter (Ludes & Anderson 1996), it was possible to evaluate any preferences expressed in relation to behavioural observations made in the presence of each litter alone, a validatory exercise considered important by Fraser et al (1993) in their discussion of the status of preference tests.
Methods
Subjects and housing
The subjects were 11 adult and juvenile white-faced capuchin monkeys, Cebus capucinus, members of an established breeding group which was housed in a series of interconnecting outdoor and indoor enclosures and rooms totaling over 230 m3. Observations were made on three wild-born adult males, two wild-born adult females, and six captive-born juveniles (three males, three females). The group's quarters were furnished with a variety of elevated walkways and resting areas, and manipulable objects. Water and food pellets (NMC, Franz Ehrle GmbH, Kehl, Germany) were available ad libitum in the smaller of the two indoor rooms. Fresh fruit and vegetables were distributed in the late afternoon once or twice a week. Further details about the group and its housing may be found in Anderson et al (1995).
Only the larger of the two indoor rooms, measuring 6.7x3.9 m, with a height of 3 m, was used for the preference tests. The two long walls of this room were coated with epoxy cement and partly tiled. The back wall, also coated in epoxy cement, contained two hatches giving access to the smaller, heated indoor room, and a transparent door which allowed technicians and researchers to move between the two indoor rooms. The front wall consisted of wire mesh with a hatch and a door giving access to the outdoor enclosures.
Procedure and statistical analysis
The floor area of the room was divided into four, equal-sized, rectangular zones (quarters) using 25 cm-high wooden planks. The planks prevented spillage of the litters into neighbouring quarters. Four litters were presented simultaneously: woodchips, wood wool, ground corn cob (as 6-8 mm gravel), and loose garden peat. The litter covered the entire floor area of each quarter. The depth of woodchips, ground corn cob and peat was between 5 and 10 cm, while some tangles of the wood wool rose above 25 cm. Further details of the litters and their general effects on the behaviour when presented individually to the monkeys are available in Ludes and Anderson (1996). Each litter was assigned randomly to a quarter and left in place for 14 consecutive days. At the end of this period, the litter was removed, the floor area was washed and allowed to dry, and then fresh litter was presented - but this time the layout of the litter locations was rotated by one quarter. This general procedure was repeated until each litter had been presented once in each quarter. Ten days of observations with each litter in each quarter were completed, giving a total of 40 days of observations. Immediately prior to preparation of the indoor room, the monkeys were ushered to the outdoor enclosure, where they were confined until preparations were finished. In order to promote initial interest in the litters each day, 140g of raisins were spread across the top of each litter immediately before each observation session. The observer (E L) then opened the hatch to allow the monkeys access to the indoor rooms and took up a seated position by the door of the back wall of the large room. Once the hatch was opened to start an observation session, the monkeys were free to move between all sectors of their enclosure.
Two, 1h observation sessions were conducted on each observation day, one in the morning and another in the afternoon. A one-zero sampling method (Martin & Bateson 1986) using intervals of 60s was used to record the following litter-related behaviours performed by any of the subjects: foraging, locomotion, social contact, and play. The behaviours were not mutually exclusive, meaning for example, that a subject moving (locomoting) across one quarter while foraging was scored as engaging in both activities. The sum of all the intervals in which at least one activity was scored was calculated post hoc to give a measure of the total presence on each litter type.
Overall scores for each litter type were obtained by summing across the four quarters. We used a Friedman analysis of variance (ANOVA) for repeated measures, followed by post hoc Wilcoxon signed ranks tests in the case of significant effects. The level of significance was set at P < 0.05.
Results
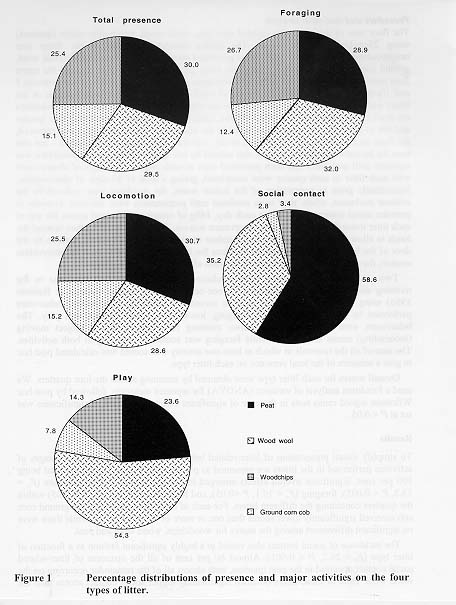
To simplify visual presentation of litter-related behavioural differences, the percentages of activities performed in the litters are presented as pie-charts in Figure 1, with the total being 100 per cent. Significant overall effects emerged in the analyses of overall presence (Fr = 13.3, P < 0.005), foraging (Fr= 10.1, P <0.05), and locomotion (Fr = 15.5, P < 0.05) within the quarters containing the different litters. For each of these three activities, the ground corn cob received significantly lower scores than one or more of the other litters, while there were no significant differences among the scores for woodchips, wood wool and peat.
The incidence of social contact also varied in a highly significant fashion as a function of litter type (Fr = 26.7, P < 0.001). Almost 60 per cent of all the instances of litter-related social contact occurred in the peat quarters, with almost all of the remainder occurring on the wood wool. Both of these litters obtained significantly higher social contact scores than the woodchips or ground corn cob. Neither the peat vs. wood wool nor the woodchips vs. ground corn cob comparisons were significant.
Similarly, there was a highly significant effect of litter type for the occurrence of play (Fr = 27.0, P <0.001), but for this activity the wood wool was significantly preferred over the other three litters. Peat was the next most frequently chosen substrate for playing in, being significantly preferred to woodchips and ground corn cob. Although play was almost twice as frequent on woodchips as compared to ground corn cob, this difference was not statistically significant.
Discussion
The results presented here show that white-faced capuchin monkeys express clear preferences when given a simultaneous choice among different floor coverings. The reactions of the monkeys to the different litters during the preference tests generally matched well with their reactions when those litters were presented individually (Ludes & Anderson 1996). This finding gives the results of the preference tests increased validity as indicators of the animals' behavioural priorities (Fraser et al 1993).
Furthermore, it seems likely that these preferences are related to physical aspects of the litters and the kinds of behaviours they promote. In terms of overall presence and locomotion, as well as foraging activity, the capuchin monkeys tended to avoid the ground corn cob. Although they did enter the ground corn cob-carpeted quarters and remove food lying on the surface soon after the start of the observation sessions, their interest in this litter quickly waned. On the other hand, the three remaining litters all held the monkeys' interest for long periods of time. Indeed, foraging activity in the wood wool, peat and woodchips clearly exceeded the availability of raisins, but the monkeys continued to sift through the litter and occasionally remove and ingest small particles for the duration of the observation periods; and supplementary informal observations indicated that foraging activity continued throughout the day. These observations reinforce the view that the environment of captive primates is enriched by increasing the opportunities for foraging with appropriate litter materials in combination with modest supplementary feeding (Anderson & Chamove 1984; Chamove & Anderson 1988; Reinhardt & Roberts 1997).
The absence of a preference for peat as a foraging substrate contrasts with the report by Ludes and Anderson (1996), in which manual foraging was over twice as common in peat as in woodchip, wood wool, or ground corn cob litters. It seems likely that methodological differences between the studies account for this difference, especially the fact that observations in the present study were restricted to the 1 hour immediately after food was scattered on all four litters, whereas observations continued for longer in the earlier study. Also, in the earlier study a mixture of dried grains was used as food, as compared with raisins in the present study; it is conceivable that food type and litter interact in ways that are yet to be identified (see also Reinhardt & Roberts 1997).
The capuchin monkeys did not spend much time in locomotion on the ground corn cob. Ludes and Anderson (1996) reported that locomotion across the floor was more frequent when the floor area was bare than when it was covered with ground corn cob litter. We reiterate the suggestion made in that report, namely that the adhesion of the small corn cob granules to the palms and the soles of the feet was perceived as unpleasant by the monkeys. It may also be the case that this type of litter has a lower absorption capacity than the others, so that increasing wetness through soiling makes the granules stick even more. Whatever the precise reasons, from the absence of positive behavioural effects in Ludes and Anderson (1996) and the relatively low scores obtained in the present choice tests, we conclude that ground corn cob granules (as a litter) do not provide effective environmental enrichment for captive capuchin monkeys. It remains to be seen to what extent this conclusion generalizes to other species of primate.
Over half of all the instances of social contact on litters occurred in the quarters containing peat. Since peat was not used disproportionately for foraging or locomotion, there must be another reason for the social contact-promoting characteristics of this litter. One important reason is communal episodes of 'peat-bathing', an activity consisting of stirring up the peat (often forming clouds of dust) and rubbing it vigorously into the pelage. No equivalent behaviour was ever observed in the other litters. Petherick and Duncan (1989) found that domestic fowl showed a preference for dustbathing in peat as opposed to simultaneously present sand, sawdust or wood shavings. These authors considered differences in coarseness, density and even taste of the substrates as possible factors underlying the preference. Descriptions and possible interpretations of the peat-bathing by capuchin monkeys, which include thermoregulation and pelage maintenance, may be found in Ludes and Anderson (1995).
Over half of all play took place in the quarters covered with wood wool. Play on this substrate was often vigorous, involving Jumping and rolling around on the 'carpet'. It seems likely that the cushioning effects of the tangled wood wool made this litter particularly suitable for play. The play-promoting properties of wood wool were also found when this was the only litter presented (Ludes & Anderson 1996).
Unlike the preferences of rodents or large herbivores, naturally arboreal monkeys' preferences for particular litter types are unlikely to be seriously affected by where the animals choose to rest or sleep. On the other hand, primates' preferences among substrates will be influenced by currently prioritized diurnal activities (as are those of other animals [Fraser et al 1993]). The extent to which primates' preferences among deep litter materials is influenced by factors such as species, experience, season and reproductive status remains to be studied, as does the issue of elasticity of demand for access to litters (Dawkins 1990). It may be concluded, however, that as in poultry and rodents (Petherick & Duncan 1989; van de Weerd et al 1996), the behavioural repertoires of captive monkeys could be enhanced by having different types of litter available in different areas of their living quarters, each promoting the expression of a particular species-specific behaviour.
Animal welfare implications
Different types of deep litter promote different types of activity in captive monkeys, including foraging, play and social contact. By using simultaneous preference tests in combination with behavioural observations, the most appropriate litters for enriching the environment of a given species or group can be identified. It may be desirable to provide more than one type of floor covering, located in different areas of the enclosure, in order to increase the range of behaviours expressed in captivity.
Acknowledgements
This research formed part of a study conducted by E Ludes-Fraulob for her doctorate from the Université Louis Pasteur, Strasbourg. The authors thank the Director and personnel of the Centre de Primatologie, Strasbourg, for help at all stages of the study.
References
Anderson J R, André E and Wolf P 1995 Successful mother- and group-rearing of a newborn capuchin monkey (Cebus capucinus) following emergency major surgery. Animal Welfare 4: 171-18
Anderson J R and Chamove A S 1984 Allowing captive primates to forage. In: Standards in Laboratory Animal Management. Part 2 pp 253-256. Universities Federation for Animal Welfare: Potters Bar, UK
Boccia M L 1989 Long-term effects of a natural foraging task on aggression and stereotypies in socially housed pigtail macaques. Laboratory Primate Newsletter 28(2): 18-19
Brent L 1992 Woodchip bedding as enrichment for captive chimpanzees in an outdoor enclosure. Animal Welfare 1: 161-170
Byrne G D and Suomi S J 1991 Effects of woodchips and buried food on behavior patterns and psychological well-being of captive rhesus monkeys. American Journal of Primatology 23: 141-151
Chamove A S and Anderson J R 1979 Woodchip litter in macaque groups. Journal of the Institute of Animal Technicians 30: 69-74
Chamove A S and Anderson J R 1988 Impact of feeding practices on growth and behavior of stump- tailed macaques (Macaca arctoides) In: Fa J E and Southwick C H (eds) Ecology and Behavior of Food-Enhanced Primate Groups pp 231-246. Alan R Liss: New York, USA
Chamove A S and Anderson J R 1989 Examining environmental enrichment. In: Segal E F (ed) Housing, Care and Psychological Well-being of Captive and Laboratory Primates pp 183-202. Noyes Publications: Park Ridge, USA
Chamove A S, Anderson J R, Morgan-Jones S C and Jones S P 1982 Deep woodchip litter: Hygiene, feeding and behavioral enhancement in eight primates species. International Journal for the Study of Animal Problems 3: 308-318
Dawkins M S 1977 Do hens suffer in battery cages? Environmental preferences and welfare. Animal Behaviour 25: 1034-1046
Dawkins M S 1983 The current status of preference tests in the assessment of animal welfare. In: Baxter S H, Baxter M R and McCormack J A D (eds) Farm Animal Housing and Welfare pp 20- 26. Martinus Nijhoff. Dordrecht, The Netherlands
Dawkins M S 1990 From an animal's point of view: Motivation, fitness, and animal welfare. Behavioral and Brain Sciences 13: 1-61
Duncan I J H 1978 The interpretation of preference tests in animal behaviour. Applied Animal Ethology 4: 197-200
Fraser D 1985 Selection of bedded and unbedded areas by pigs in relation to environmental temperature and behaviour. Applied Animal Behaviour Science 14: 117-126
Fraser D, Phillips P A and Thompson B K 1993 Environmental preference testing to assess the well- being of animals - an evolving paradigm. Journal of Agricultural & Environmental Ethics 6 (Supplement 2): 104-114
Hughes B O and Black A J 1973 The preference of domestic hens for different types of battery cage floor. British Poultry Science 14: 615-619
Ludes E and Anderson J R 1995 'Peat-bathing' by captive white-faced capuchin monkeys (Cebus capucinus). Folia Primatologica 65: 38-42
Ludes E and Anderson J R 1996 Comparison of the behaviour of captive white-faced capuchin monkeys (Cebus capucinus) in the presence of four kinds of deep litter. Applied Animal Behaviour Science 49: 293-303
Manser C E, Morris T H and Broom D M 1994 Do rats prefer solid or gridded floors? In: Bunyan J (ed) Welfare and Science pp 462-463. Royal Society of Medicine Press: London, UK
Martin P and Bateson P 1986 Measuring Behaviour. Cambridge University Press: Cambridge, UK
Mench J A 1994 Environmental enrichment and exploration. Lab Animal 23(2): 38-41
Mulder J B 1975 Bedding preferences of pregnant laboratory-reared mice. Behavior Research Methods and Instrumentation 7: 21-22
Natzke R P, Bary D R and Everett R W 1982 Cow preference for free stall surface material. Journal of Dairy Science 65: 146
Petherick J C and Duncan I J H 1989 Behaviour of young domestic fowl directed towards different substrates. British Poultry Science 30: 229-238
Poole T B 1988. Behaviour, housing and welfare of non-human primates. In: Beynen A C and Solleveld HA (eds) New Developments in Biosciences: Their Implications for Laboratory Animal Science pp 231-237. Martinus Nijhoff: Dordrecht, The Netherlands
Reinhardt V and Roberts A 1997 Effective feeding enrichment for non-human primates: A brief review. Animal Welfare 6: 265-272
Rosenblum L A and Smiley J 1984 Therapeutic effects of an imposed foraging task in disturbed monkeys. Journal of Child Psychology and Psychiatry 25: 485-497
van de Weerd H A, van Loo P L P, van Zutphen L F M, Koolhaas J M and Baumans V 1997 Preferences for nesting material as environmental enrichment for laboratory mice. Laboratory Animals 31: 133-143
This article originally appeared in Animal Welfare 8, 127-134 (1999).
Reprinted with permission of the Editor.